Unconventional thinking for value that goes beyond contract manufacturing


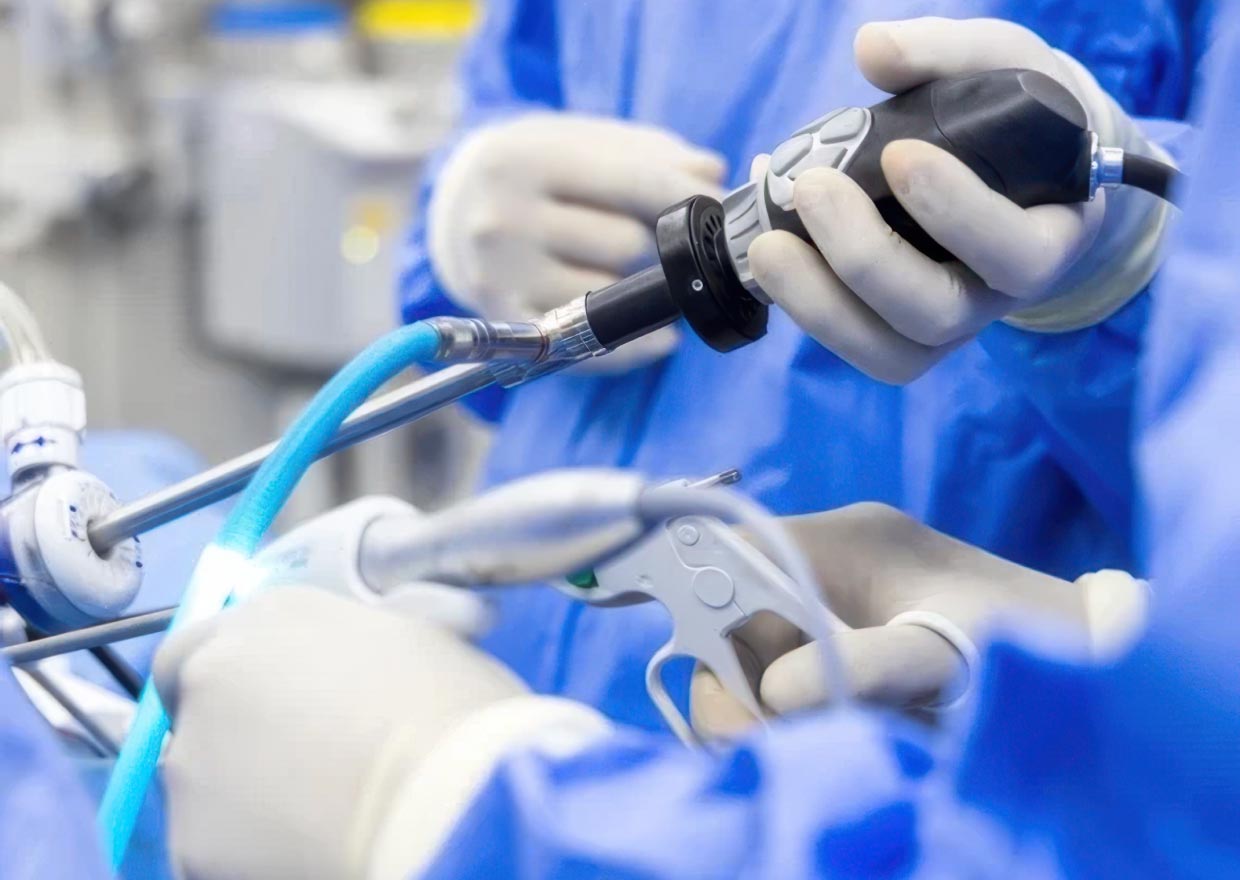
Schivo is a leading contract manufacturer of medical device components, sub-assemblies, and assemblies. A specialist in minimally invasive, robotic-assisted, cardiology, and orthopedic surgery systems, our unconventional thinking drives dramatic improvements in OEM productivity, quality, and speed to market. We provide value that goes beyond manufacturing to optimize OEM profit and enhance the patient experience.
We are unbound by traditional methodologies or approaches
Schivo’s non-traditional thinking exceeds the limitations of conventional methods to help ensure the best cost and elimination of risk across the contract manufacturing process. We do not accept the limitations of customary solutions and encourage unorthodox thinking to unlock hidden value in the supply-to-point-of-care chain. Our strong process expertise helps anticipate problems before they become issues and identifies better ways of working. Plus, our vertical integration reduces cost and accelerates product development and manufacturing time.
Schivo’s ISO 13485-certified and FDA-registered facilities support the world’s leading medical device OEMs. We provide a project financial structure that ensures a competitive product and assures patients receive an effective device or procedure that insurers will reimburse.
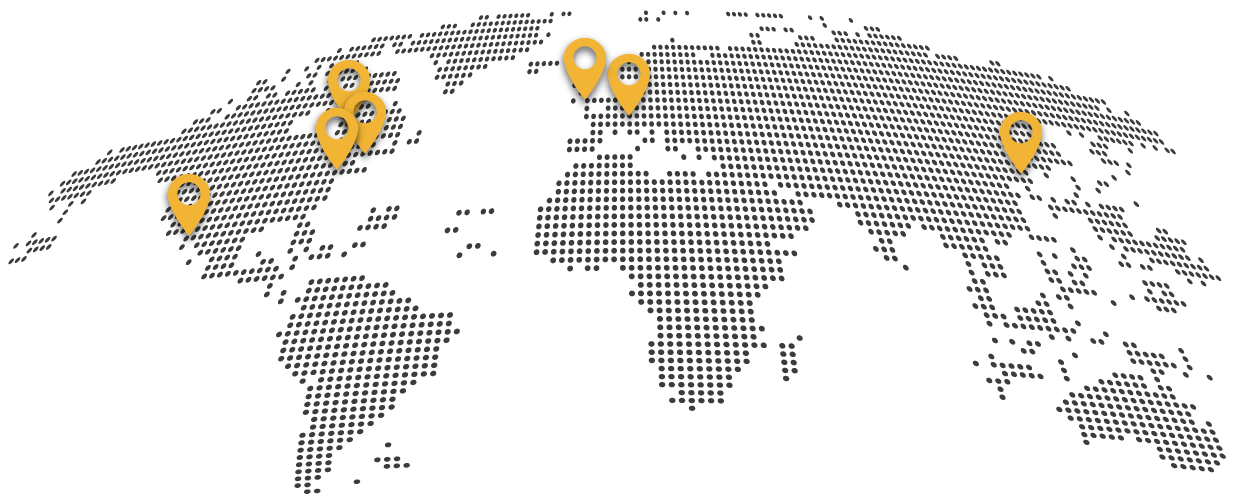
A member of the global Schivo Group
Schivo is part of the Schivo Group, a global leader in high-precision manufacturing for the medical device, analytical instrument, clinical diagnostic, and genomics markets. Based in Waterford, Ireland, the company has more than 630 employees at facilities in Europe, North America, and Asia.